Doyle-Fuller-Newman (DFN) battery model
The DFN model, also called P2D or the Newman model, is the canonical physics-based model for a lithium-ion cell. Full porous electrode theory. Spatially resolved transport. The benchmark against which every reduced-order model is compared.
DFN is the right model when your engineering question depends on where something happens inside the electrode: lithium plating near the separator, gradients in graded architectures, mechanism-level degradation. Reduced-order SPM and SPMe models are faster but cannot resolve those gradients.
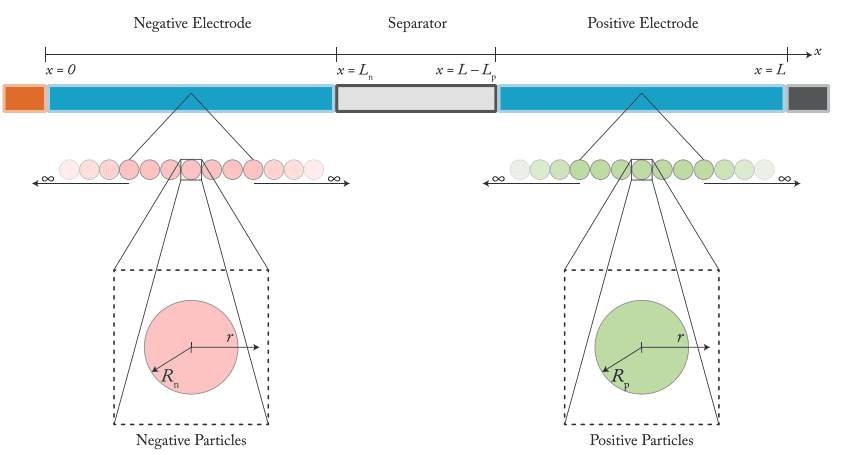
DFN model structure: porous electrodes, electrolyte transport, particle diffusion
What DFN captures
DFN implements full porous electrode theory. Both electrodes are treated as porous media with spatially resolved transport in the electrolyte and solid phases. Intercalation kinetics, charge conservation, and species transport are coupled across a 1D domain spanning the anode, separator, and cathode. Inside each electrode, a "pseudo" radial dimension inside the particles tracks solid-state lithium diffusion (this is why the model is also called P2D).
The model is invoked in PyBaMM as pybamm.lithium_ion.DFN(). It serves two roles in practice: it is the working model for studies that need spatial physics, and it is the reference model that reduced-order SPM and SPMe are validated against.
Inside the model
Porous electrodes
Spatially resolved transport
Both electrodes are treated as porous media. Lithium concentration in the electrolyte and in the solid phase varies through the thickness of the electrode stack. The model resolves what happens at the separator versus at the current collector.
Electrolyte transport
Concentration and potential gradients
Concentrated solution theory tracks lithium-ion concentration and electrolyte potential across the anode, separator, and cathode. Transport limitations and ohmic drops are observable, not lumped.
Solid diffusion
Per-electrode particle physics
Each electrode has its own particle population, intercalation kinetics, and OCV curve. Anode and cathode contributions to the cell voltage are separable, and electrode-specific overpotentials are inspectable.
Coupled physics
Reference for reduced-order models
DFN is the benchmark against which SPM and SPMe are compared. Every PyBaMM degradation submodel (SEI, plating, LAM, particle cracking) attaches to the DFN framework.
Spatial resolution: the differentiator
The defining advantage of DFN over reduced-order models is spatial resolution within the electrode. Anode potential is not uniform across the electrode thickness. Near the separator, where lithium flux peaks during charging, the anode potential drops lower than near the current collector. Plating risk is highest in that region, and DFN captures the gradient that determines whether plating occurs and where. SPM and SPMe average over the electrode thickness and cannot see this.
The same principle applies to non-uniform porosity in graded electrodes, to reaction-rate distributions that shift with C-rate and temperature, and to electrolyte depletion profiles that vary spatially through the stack. If your question involves where something happens within the electrode, not just whether it happens, DFN is required.
DFN at a glance
| Dimension | DFN |
|---|---|
| Physics captured | Full porous electrode theory + electrolyte transport |
| Parameterization burden | High (35+ parameters, teardown + half-cell typical) |
| Compute cost | High (coupled PDEs, expensive for sweeps) |
| Accuracy | Benchmark for lithium-ion cell models |
| Degradation capability | Full mechanism-level (SEI, plating, cracking, LAM) |
| Best fit | Plating studies, design optimization, fast charge, low temperature |
For a side-by-side comparison with ECM, SPM, and SPMe, see the electrochemical model types resource.
When DFN is the right model
DFN earns its parameterization and compute cost when the engineering question depends on spatial physics. Three patterns recur.
Fast-charge envelope
Anode potential drops below 0 V vs. Li/Li⁺ near the separator first. DFN resolves that gradient, which is what determines plating onset and where it occurs.
Electrode design
Coating thickness, porosity profiles, particle size distributions, and graded architectures all change cell behavior through spatial mechanisms that SPM and SPMe average away.
Mechanism-level degradation
SEI growth on freshly cracked surfaces, position-dependent plating, electrolyte depletion profiles. Each of these is spatially distributed inside the electrode.
Where DFN is overkill: low-rate calendar studies, first-pass design sweeps over hundreds of points, real-time onboard estimation in a BMS. Those are SPM, SPMe, or ECM territory. The right rule of thumb: start with the lowest-fidelity model that answers the question, and step up only when the physics demand it.
Beyond standard 1D DFN
Standard DFN assumes a single active material per electrode and a monotonic OCV curve. Several important chemistries and cell geometries violate those assumptions.
Composite electrodes. Graphite-silicon blends are the most common case. PyBaMM's BasicDFNComposite models each active material phase with its own particle diffusion and OCV curve, capturing the differential lithiation between graphite and silicon domains.
Hysteresis. LFP and silicon chemistries exhibit OCV hysteresis: the voltage on charge differs from the voltage on discharge at the same SoC. PyBaMM includes hysteresis state models that track the charge/discharge branch and matter for SoC estimation accuracy and for path-dependent degradation prediction.
MSMR thermodynamics. The Multi-Species Multi-Reaction framework (Yang et al. 2017) models multiple intercalation reactions within a single electrode particle, capturing graphite staging plateaus and NMC phase transitions with higher thermodynamic accuracy than a fitted polynomial OCV.
3D extension. Large-format pouch and prismatic cells have in-plane gradients that 1D DFN cannot see. Tab placement affects internal resistance distribution; temperature hotspots localize near tabs. Pseudo-3D and full 3D electrochemical models resolve these gradients without rewriting the parameter set.
Parameterization is the bottleneck
DFN parameterization is the heaviest in the model hierarchy. Half-cell OCV curves for both electrodes. A full-cell OCV characterization. Geometric parameters from teardown: electrode dimensions, particle sizes, porosities, active material volume fractions. Rate capability tests to fit transport and kinetic properties as functions of temperature and SoC. Chen et al. (2020) demonstrated 35+ parameters for a single LG M50 21700 cell.
A solver runs in seconds. Assembling a defensible parameter set often takes weeks. Identifiability is harder than fit quality: a parameterization that pushes correlated parameters in opposite directions can fit one curve perfectly and fail on the next one. Our parameter estimation guide covers the DFN data requirements step by step.
This is the Train stage in Ionworks Studio. A DFN parameter set fit here drives every downstream study, including 3D electrochemical simulations of the same cell, without re-fitting.
How Ionworks fits in
How Ionworks supports DFN
01
PyBaMM as the DFN solver
Ionworks runs DFN through PyBaMM (`pybamm.lithium_ion.DFN()`), the open-source framework maintained by the Ionworks team. The same parameter set drives DFN, SPMe, and SPM, so the model choice is a setting in the workflow, not a re-parameterization.
02
Parameterization with provenance
A 35+ parameter set is too large to manage by spreadsheet. Each parameter is recorded with its source: which experiment it was fit to, which literature reference it came from, which cell it belongs to. Studies six months later are reproducible because the provenance travels with the parameter set.
03
Identifiability and uncertainty
Global optimization with multistart, Bayesian priors, and uncertainty quantification flag parameters that the data leaves under-constrained. A defensible parameter set is more useful than a precise-looking one.
04
BPX import and export
Ionworks supports BPX, the community standard for physics-based battery parameter sets. Vendor-supplied BPX files load directly. Internal parameter sets export to BPX for sharing with partners and suppliers without translation.
05
1D to 3D without rewriting
A DFN parameter set fit on cycling data carries forward into 3D electrochemical simulations of the same cell. Tab placement, hotspots, and in-plane gradients are studied without maintaining a separate parameter set in a different tool.
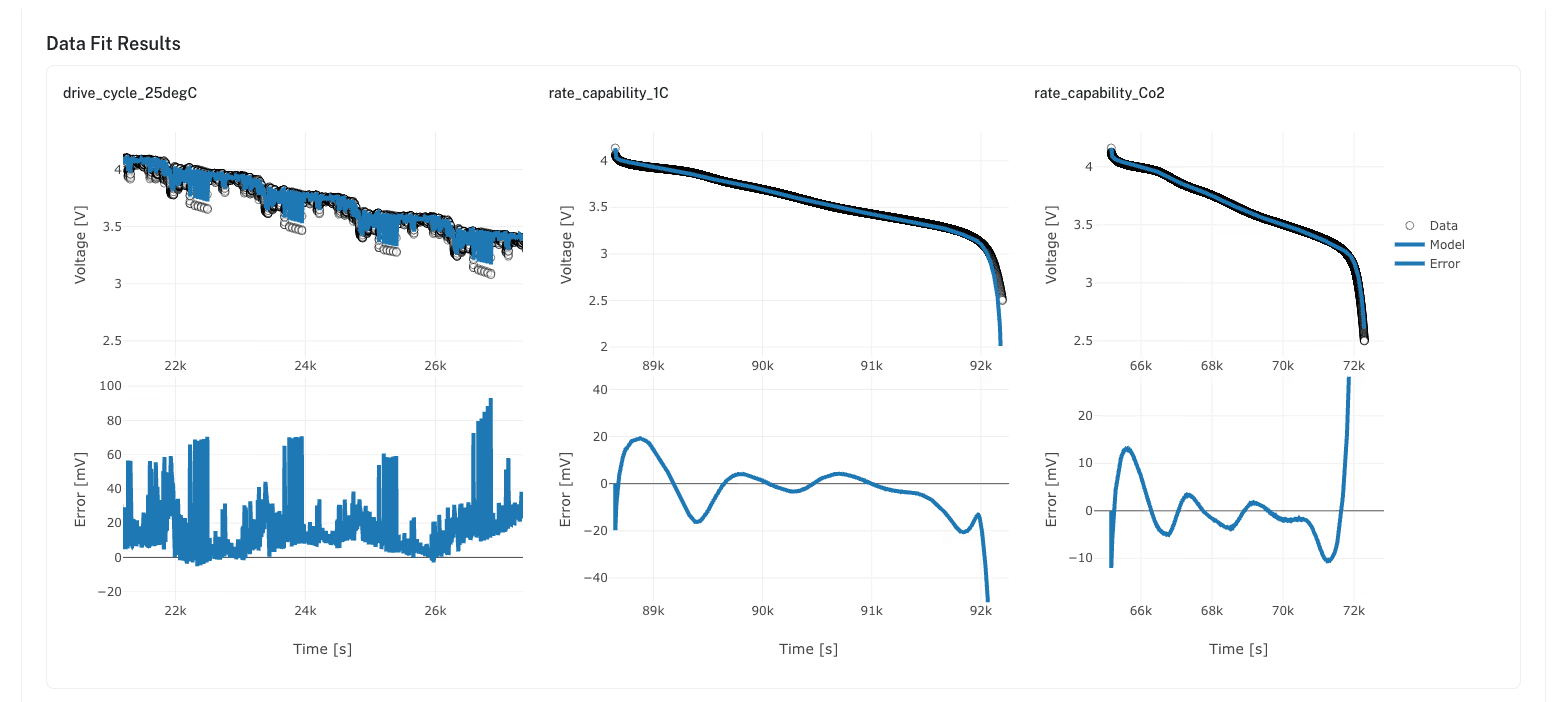
DFN parameterization in Ionworks Studio
Frequently asked questions
pybamm.lithium_ion.DFN(). Some papers use one term, some use the other, and they are interchangeable in the literature.BasicDFNComposite), MSMR thermodynamics, and OCV hysteresis variants are all supported.See DFN running on your cell, with full parameter provenance
Bring a dataset and a parameter set, or start from one of ours. We will show what DFN catches that SPMe averages away, and where the cost pays for itself.